Sublimation
Examples of sublimation
As mentioned above, (CO2) is a common example of a chemical compound that sublimates at atmospheric pressure—a block of solid CO2 at room temperature and one atmosphere pressure will turn into gas without first becoming a liquid. Iodine is another substance that visibly sublimates at room temperature. In contrast to CO2, though, it is possible to obtain liquid iodine at atmospheric pressure by heating it.
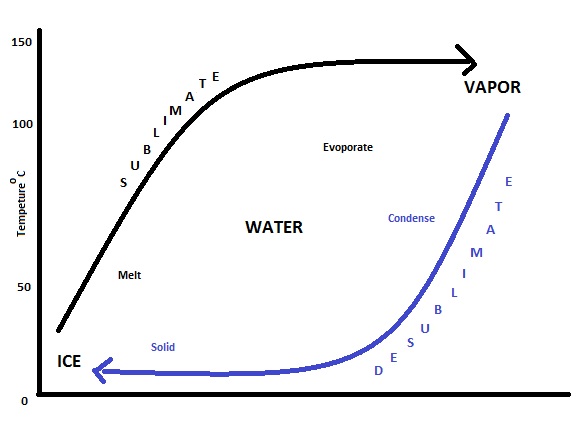
Principles of sublimation
Sublimation is a phase transition that occurs at temperatures and pressures below what is called the "triple point" of the substance (see phase diagram). The process is an endothermic change—that is, a change in which heat is absorbed by the system. The enthalpy of sublimation can be calculated as the enthalpy of fusion plus the enthalpy of vaporization.
